 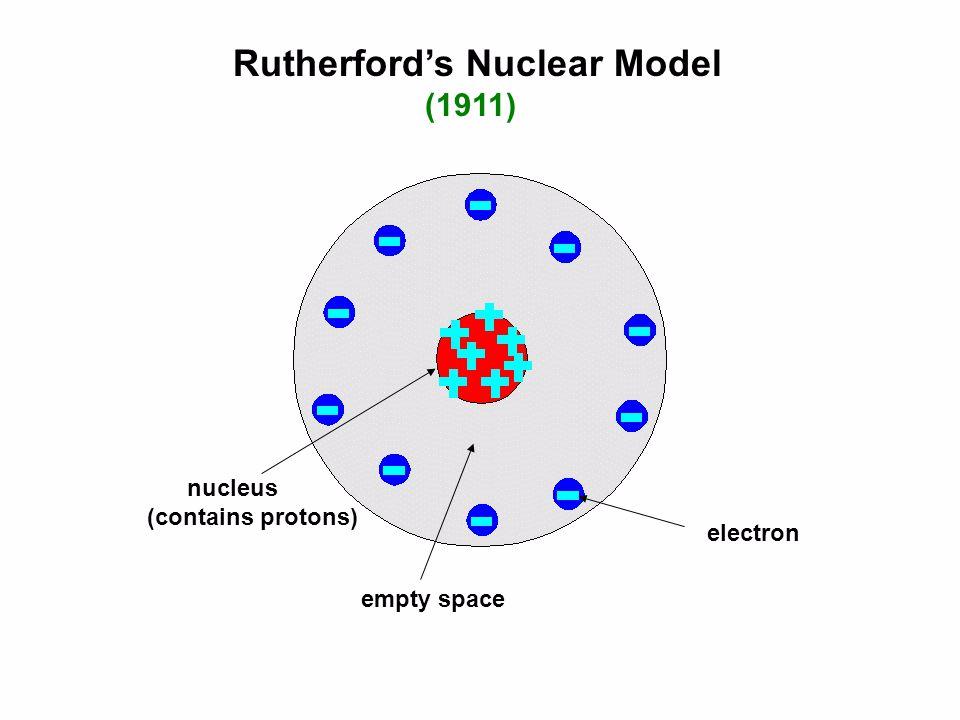
It challenged the previous notion of the atom being a solid, indivisible entity and proposed a more complex organization. Rutherford’s model of the atom, known as the planetary model, provided a new understanding of atomic structure. This nucleus contained most of the atom’s mass and was surrounded by orbiting electrons, much like planets revolving around the sun. Through his experiments, Rutherford found that atoms consisted mostly of empty space, with a small, dense, and positively charged central nucleus. Rutherford’s Discovery of the Atomic NucleusĮrnest Rutherford, the renowned physicist, made a groundbreaking discovery that revolutionized our understanding of atomic structure – the existence of the atomic nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
